Ecuador's healthcare landscape is uniquely characterized by its geographical diversity, where coastal humidity and high-altitude Andean climates dictate the specific breathability and barrier requirements for ppe gowns. The demand for reliable protection is constant across public and private sectors to prevent cross-contamination in densely populated urban centers like Quito and Guayaquil.
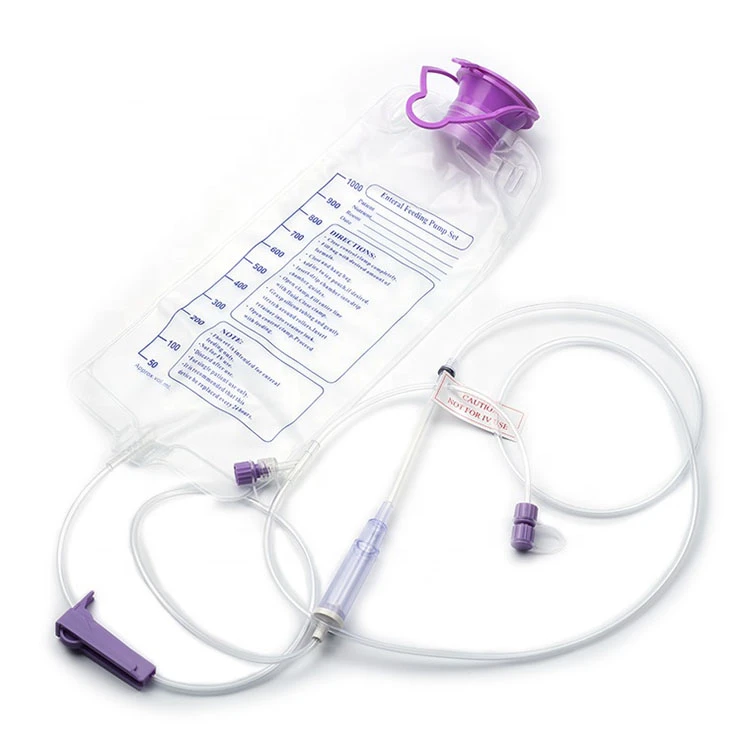
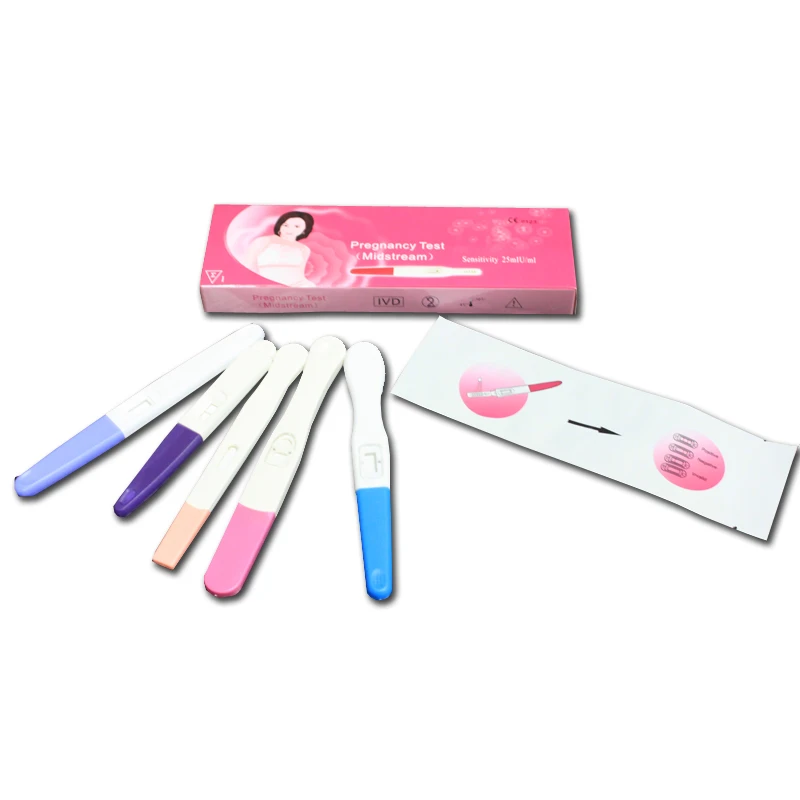
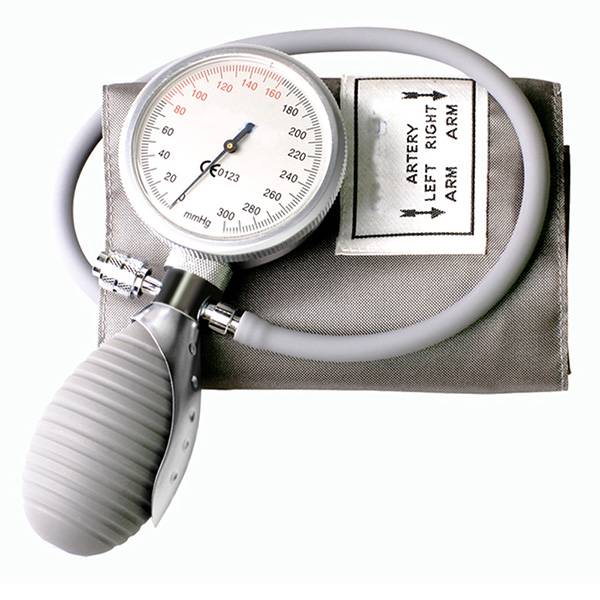
Currently, the market is shifting from basic protective wear to highly specialized medical ppe kit solutions. This transition is driven by a need for standardized safety protocols that can be deployed rapidly in both state-run hospitals and remote rural clinics, where logistics often complicate the supply of sterile consumables.
Economic fluctuations in the region have led to a strategic focus on cost-effective yet high-quality disposable options. The adoption of advanced non-woven fabrics ensures that clinicians maintain a rigorous aseptic environment without compromising the efficiency of their workflows in high-pressure medical settings.