Edta K2 K3 Edta Tubes Edta K2 Or K3 Tube is a key solution in the medical device industry, specifically within In vitro diagnostic equipment and blood collection tube. This article explores how HEBEI ORIENT IMPORT & EXPORT CO., LTD supports professionals with durable, high-performance products, and explains why this product is an ideal choice for businesses in these sectors.
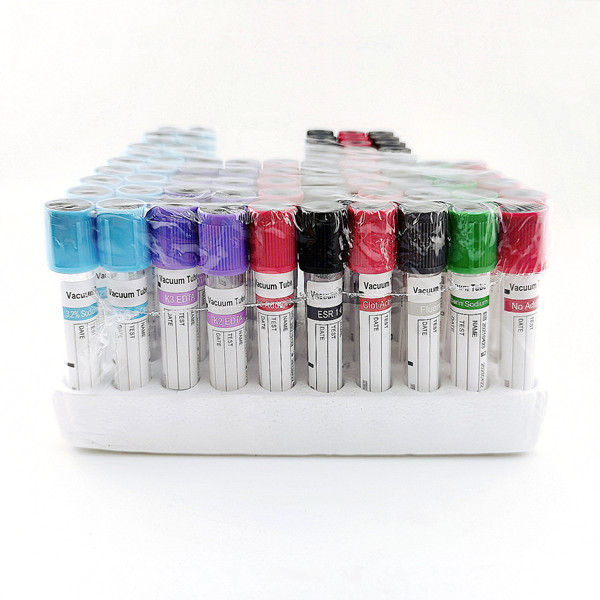
Table of Contents
- Edta K2 K3 Edta Tubes Edta K2 Or K3 Tube Overview
- Benefits & Use Cases of Edta K2 K3 Edta Tubes Edta K2 Or K3 Tube in blood collection tube
- Cost, Maintenance & User Experience
- Sustainability & Market Trends in medical device
- Conclusion on Edta K2 K3 Edta Tubes Edta K2 Or K3 Tube from HEBEI ORIENT IMPORT & EXPORT CO., LTD
Edta K2 K3 Edta Tubes Edta K2 Or K3 Tube Overview
In hematology and routine diagnostics, anticoagulation accuracy is essential. The Edta K2 K3 Edta Tubes Edta K2 Or K3 Tube family provides reliable whole-blood preservation through K2EDTA (dipotassium) or K3EDTA (tripotassium) additives that chelate calcium to prevent clotting. These lavender-cap tubes are widely used for CBC, HbA1c, blood grouping workflows, and select molecular applications where cell integrity and nucleic acid stability are critical. For B2B buyers standardizing IVD consumables, consistent vacuum accuracy, additive distribution, and stopper integrity directly affect throughput and repeat testing rates.
- Available in common formats (e.g., 13×75 mm and 13×100 mm) with typical draw volumes from ~2 mL to ~6 mL to fit automated analyzers and phlebotomy routines.
- K2EDTA options are usually spray-dried for minimal dilution; K3EDTA can be liquid or spray-dried depending on configuration, supporting morphology-sensitive assays.
- Manufactured as sterile, single-use tubes with color-coded closures and lot traceability—aligned with widely recognized IVD quality practices and dimensional standards for automated lines.
- Backed by HEBEI ORIENT IMPORT & EXPORT CO., LTD, a reliable partner offering stable supply, OEM/ODM options, and responsive service for hospital, lab, and distributor networks.
Benefits & Use Cases of Edta K2 K3 Edta Tubes Edta K2 Or K3 Tube in blood collection tube
In the blood collection tube category, K2 and K3 EDTA formats support core workflows: complete blood count (CBC), WBC/RBC/platelet indices, blood bank screening, HbA1c, and selected molecular assays. Many laboratories prefer K2EDTA for routine hematology due to lower sample dilution and preserved cell morphology, while K3EDTA may be chosen for specific analyzer protocols. These tubes are engineered for consistent additive coating, secure sealing, and compatibility with automated decappers and transport systems—helping minimize pre-analytical variability that can trigger redraws.
- Applications: CBC and differential, reticulocyte counts, blood grouping workflows, and HbA1c; not intended for calcium- or metal-sensitive chemistries where chelation interferes with results.
- Competitive advantages: accurate vacuum draw, reliable stopper reseal, uniform EDTA distribution, clear labeling areas/barcode-ready sleeves, and options for custom branding.
- HEBEI ORIENT IMPORT & EXPORT CO., LTD brings category expertise—supporting product selection (K2 vs K3), size harmonization across analyzers, and packaging configurations for central labs and decentralized sites.

Cost, Maintenance & User Experience
Total cost of ownership for EDTA tubes extends beyond unit price. With Edta K2 K3 Edta Tubes Edta K2 Or K3 Tube, buyers gain consistency that helps reduce repeat sampling, manual interventions, and analyzer alarms—hidden costs that impact KPIs. Durable packaging limits in-transit damage; long, clearly marked shelf life supports bulk procurement without excess expiry waste. For high-volume labs, dimensional consistency improves automation uptime, and reliable vacuum draw reduces underfill and clot-risk events when the IFU is followed.
- ROI drivers: fewer redraws, stable specimen quality within validated storage intervals, and smoother integration with conveyors, decappers, and hematology analyzers.
- User feedback from IVD settings often highlights predictable fill volumes, secure capping, and easy visual inspection—small details that streamline phlebotomy and bench workflows.
Sustainability & Market Trends in medical device
The IVD consumables market is moving toward safer materials, transparent traceability, and waste reduction. Procurement teams increasingly request lighter components, optimized secondary packaging, and clear instructions for responsible disposal. Regulatory focus—such as EU IVDR labeling, UDI traceability, and adherence to recognized IVD quality systems—continues to shape specifications for blood collection tubes. Many laboratories are also standardizing on K2EDTA for hematology to minimize dilution effects while maintaining morphology, aligning with analyzer manufacturer preferences.
- HEBEI ORIENT IMPORT & EXPORT CO., LTD supports forward-looking purchasing with stable quality, clear product documentation, and options that help reduce transport and storage footprints.
- By aligning with widely recognized practices for IVD manufacturing and tube dimensions, the company helps partners meet evolving compliance requirements while enhancing operational efficiency.
Conclusion on Edta K2 K3 Edta Tubes Edta K2 Or K3 Tube from HEBEI ORIENT IMPORT & EXPORT CO., LTD
For laboratories and distributors aiming to optimize pre-analytical quality, the Edta K2 K3 Edta Tubes Edta K2 Or K3 Tube line delivers dependable anticoagulation, analyzer compatibility, and procurement-friendly options. As a trusted manufacturer-partner, HEBEI ORIENT IMPORT & EXPORT CO., LTD offers consistent quality, competitive supply, and OEM support to help standardize your hematology workflows at scale.
- Contact us: email: info@cnort.com
- Visit our website: https://www.orientmedicare.com
Nov . 19, 2025 14:42