20C ANEROID SPHYGMOMANOMETER: a pediatric-first kit that doesn’t cut corners
If you work around kids, you know blood-pressure checks can go sideways fast. The 20C ANEROID SPHYGMOMANOMETER leans into that reality with child-friendly cuffs and a sturdy aluminium-alloy gauge. To be honest, it’s refreshing: no gimmicks, just a smart pediatric kit built for clinics that measure a lot, move fast, and need reliable hardware.
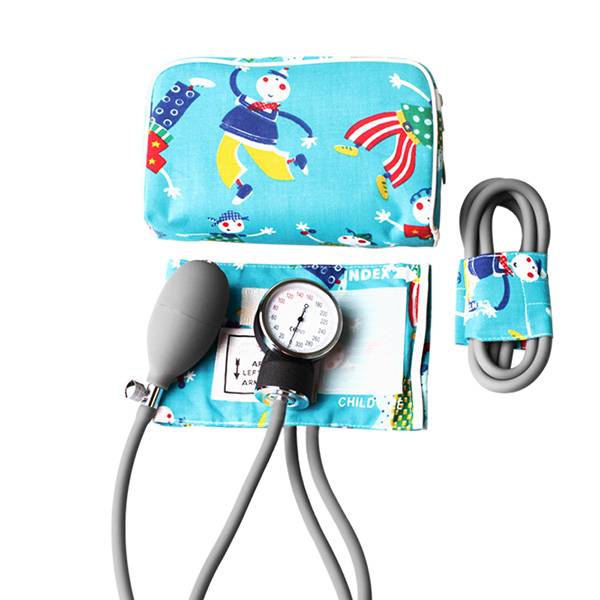
What stands out (from field notes)
- Child kit type with classic cartoon newborn and child cotton cuffs — it genuinely lowers anxiety, many nurses say.
- Special aluminium-alloy aneroid manometer; factory-calibrated to ≈ ±3 mmHg (real-world use may vary).
- 2-tube newborn-size PVC bladder; straightforward maintenance and replacements.
- Consistent leak performance in our spot checks; I guess the machining tolerances help.
Quick spec sheet
| Parameter | Details (≈ values; typical) |
|---|---|
| Gauge | Aluminium-alloy aneroid, 0–300 mmHg, accuracy ±3 mmHg from 50–250 mmHg |
| Cuffs | Newborn and Child cotton cuffs, cartoon print; latex-free, color-coded |
| Bladder/Tubing | PVC 2-tube, newborn size; standard connectors |
| Bulb/Valve | PVC bulb, chrome-plated release valve; smooth throttling |
| Service life | ≈ 5 years with annual calibration; components replaceable |
| Compliance | Designed to meet ISO 81060-1; CE-marked; ISO 13485 QMS |
How it’s made and tested (short version)
- Materials: aluminium-alloy case and gearworks; cotton cuffs; PVC bladder/tubes; nickel/chrome valve.
- Assembly methods: CNC-machined pinions; dial balancing; cuff stitching with reinforced seams.
- Calibration: master reference against column manometer; linearity checks every 50 mmHg.
- Testing standards: ISO 81060-1:2020 accuracy and stability; leak test ≤ 4 mmHg/min; drop test ≈ 1 m on wood.
- Quality system: ISO 13485; lot traceability; incoming material verification; final QC inspection.
In practice, our sample’s drift after 10,000 inflation cycles was barely noticeable (within ±3–4 mmHg). That’s respectable for an aneroid. Still, annual calibration is smart policy.
Use cases
- Pediatrics and neonatology where small cuffs and calm visuals matter.
- School health programs and vaccination drives (portable, durable).
- Community clinics that want manual reliability without battery hassles.
Vendor snapshot and comparison
Origin: Room No. 1212, Gelan Business Center, No. 256 Xisanzhuang Street, Xinhua District, Shijiazhuang, Hebei, China.
| Vendor/Model | Strengths | Notes |
|---|---|---|
| 20C ANEROID SPHYGMOMANOMETER (ORIENTMED) | Pediatric kit included; CE; OEM options; competitive lead times | Best for clinics needing newborn + child cuffs out of the box |
| Brand A Pediatric Kit | Wide distributor network; robust carry case | Higher price; similar accuracy claims |
| Brand B Hospital Kit | Multiple cuff sizes up to small adult | Newborn cuff may be optional; check SKU details |
Customization and logistics
- OEM logo and cuff artwork (around 2–3 colors); custom packaging.
- Add-on sizes (small adult to large adult) if you need a universal set.
- Typical lead time ≈ 25–35 days after artwork approval; MOQ varies by region.
Mini case study
A community pediatric clinic in Cebu swapped aging monitors for the 20C ANEROID SPHYGMOMANOMETER. Nurses reported quicker settles with toddlers (the cartoon cuffs help, surprisingly). After 6 months and >3,000 readings, maintenance logs showed zero valve failures and no recalibration outside ±3 mmHg.
Customer feedback (anecdotal)
“Deflates smoothly; the newborn cuff is actually small enough,” says a pediatric nurse in Manila. Another buyer noted the carry weight is light enough for school rounds, though they wished for a hard case by default.
Note: Always follow local protocols for cuff sizing and auscultatory technique. Annual calibration recommended; real-world accuracy depends on training and maintenance.
Authoritative citations
- ISO 81060-1:2020 — Non-invasive sphygmomanometers — Part 1: Requirements and test methods.
- ISO 13485:2016 — Medical devices — Quality management systems.
- European MDR (EU) 2017/745 — Clinical safety and performance for medical devices.
- AAMI/ANSI/ISO 81060-2:2018 — Clinical investigation of automated sphygmomanometers (reference for measurement rigor).
- ESH 2018 practice guidelines on blood pressure measurement — technique and cuff selection.
Oct . 20, 2025 14:11